Immunological health: the role of EPHB3 in enabling and preserving human T-cell immunity
Prinicipal Investigator

Prof. Dr. Horst von Bernuth
Scientific interest within the context of the graduate college
Health is not merely the absence of disease, but a dynamic, active molecular process where the development and preservation of a competent immune system is crucial. A core characteristic of immunological health is thymopoiesis. Impaired development or decay of thymopoietic capacity shorten the health span through increased susceptibility to infectious diseases, cancer, autoimmunity, and inflammatory disorders.1,2 To extend the human health span, we need to define the homeostatic pathways that actively enable and preserve immunological health. Still, crucial signaling pathways governing T-cell development in the thymus are insufficiently characterized on the molecular level in humans. Such insights are important for the early detection of deviations from health for diagnostic purposes and for the development of intervention strategies to re-establish healthy thymopoiesis.1,3
Patients with rare mutations in key pathways for the development and function of both the thymus stromal compartment and the lymphoid compartment offer a unique model to address this knowledge gap. We aim to identify the health-enabling signaling networks required to build and preserve thymus-dependent T-cell homeostasis as a foundation for future diagnostics and interceptive therapies to establishing and maintaining immunological health.
Project description
This project aims at defining the functional contribution of EPHB3 to immunological health, using a newly identified mutation in combined immunodeficiency (CID) patients. EPHB3 is a Receptor Tyrosine Kinase (RTK) capable of bidirectional signaling.4 While murine knock-out models (Ephb3−/−) demonstrate T-cell lymphopenia and premature immunosenescence as a consequence of impaired thymopoiesis, its role in human remains unknown.5 We have identified two patients from a single family (mother and son) presenting with a variant in the ligand-binding domain (LBD) of the EPHB3 gene. One patient exhibits autoimmunity with early-onset juvenile idiopathic arthritis and there is infection susceptibility in the family history with even an early fatal pneumonia, mirroring the murine phenotype and indicating dysfunctional thymopoiesis. Accordingly, the immunological phenotype displays a reduction in T-, B- and NK cells and thus, is classified as CID. This represents the first-in-human description of a disease-causing variant in EPHB3, offering the unique opportunity to study the role of EPHB3 in human immunological health. According to available data, EPHB3 is expressed within thymic epithelial cells (TECs), suggesting a critical role in the thymic stroma.6 However, our own preliminary data using artificial thymic organoids (ATOs) in vitro display an additional cell-intrinsic defect in developing T-cell precursors, pointing towards a previously not described, T-cell intrinsic function of EPHB3. We hypothesize that the variant impairs ligand binding and signal transduction, thereby disrupting the critical ligand-receptor crosstalk required for development of both thymic stroma cells and T-cells.
Therefore, the MD candidate will assess the functional role of EPHB3 for both thymic stroma cells as well as T-cell precursors and characterize the impact of the newly identified mutation (Figure 1). Specifically, in work package 1 (WP1), the candidate will conduct a functional validation of the mutation (by RT-PCR, Western blotting, and phosphorylation assays) using primary patient samples (PBMCs) as well as edited cell lines generated by the candidate (Ephb3−/− HEK 293T cell line with overexpression plasmids coding for wild type vs. patient variant). In WP2, the candidate will assess the functional contribution of EPHB3 for T lymphocytes maturation and functionality by transcriptomic (RNAseq) and phenotypic (FACS) profiling of patient-derived and control T-cell subsets ex vivo as well as of patient and control HSC-derived T-cell precursors generated in ATOs in vitro.7 To assess the role of EPHB3 for thymic stroma cells (WP3), the candidate will leverage an in vitro iPSC to thymus epithelial progenitor cell differentiation protocol. The required (patient-derived) iPSC lines, will be generated by the BIH Core Unit pluripotent Stem Cells and Organoids (CUSCO).8,9 To assess a possible role of EPHB3 for premature immunosenescence of T-cells (WP4), the candidate will conduct profiling (epigenetic, genomic, and phenotypic) for markers of cellular senescence in primary T cells from the patients and healthy controls.10,11 The candidate will also assess autoantibodies in the sera of patients and controls, as an indicators of premature thymic involution.12,13,14 Embedded in the Else Kröner-Promotionskolleg Re-Thinking Health, this project enables a first-in-human description of the function of EPHB3 for immunological health with future translational capacity.
Abbreviations: HSCs, hematopoietic stem cells; LBD, ephrin ligand-binding domain; WP, work package
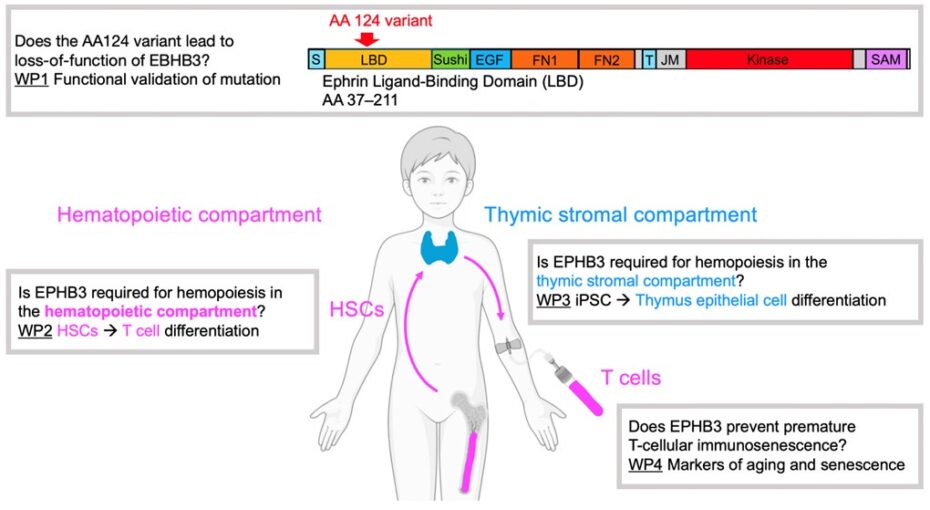
Figure 1. Visual project summary.
References
- Dinges SS, Amini K, Notarangelo LD, Delmonte OM. Primary and secondary defects of the thymus. Immunol Rev. 2024; 322(1): 178-211.
- Liu Z, Liang Q, Ren Y, Guo C, Ge X, Wang L, Cheng Q, Luo P, Zhang Y, Han X. Immunosenescence: molecular mechanisms and diseases. Signal Transduct Target Ther. 2023; 8(1): 200.
- Kreins AY, Maio S, Dhalla F. Inborn errors of thymic stromal cell development and function. Semin Immunopathol. 2021; 43(1): 85-100.
- Pasquale EB. Eph receptor signaling complexes in the plasma membrane. Trends Biochem Sci. 2024; 49(12): 1079-1096.
- García-Ceca J, Montero-Herradón S, Zapata AG. Thymus aging in mice deficient in either EphB2 or EphB3, two master regulators of thymic epithelium development. Dev Dyn. 2020; 249(10): 1243-1258.
- Speir ML, Bhaduri A, Markov NS, Moreno P, Nowakowski TJ, Papatheodorou I, Pollen AA, Raney BJ, Seninge L, Kent WJ, Haeussler M. UCSC cell browser: visualize your single-cell data. Bioinformatics. 2021; 37(23): 4578-4580.
- Bosticardo M, Pala F, Calzoni E, Delmonte OM, Dobbs K, Gardner CL, Sacchetti N, Kawai T, Garabedian EK, Draper D, Bergerson JRE, DeRavin SS, Freeman AF, Güngör T, Hartog N, Holland SM, Kohn DB, Malech HL, Markert ML, Weinacht KG, Villa A, Seet CS, Montel-Hagen A, Crooks GM, Notarangelo LD. Artificial thymic organoids represent a reliable tool to study T-cell differentiation in patients with severe T-cell lymphopenia. Blood Adv. 2020; 4(12): 2611-2616.
- Provin N, d’Arco M, Le Bozec A, Kervagoret E, Bruneau A, Brusselle L, Fourgeux C, Poschmann J, Hulin P, Maminirina P, Baron O, Saulquin X, Guillonneau C, David L, Giraud M. Combinatory differentiation of human induced pluripotent stem cells generates functional thymic epithelium driving dendritic cell and CD4/CD8 T-cell development. Nat Commun. 2026; 17(1): 1969.
- Yamazaki Y, Urrutia R, Franco LM, Giliani S, Zhang K, Alazami AM, Dobbs AK, Masneri S, Joshi A, Otaizo-Carrasquero F, Myers TG, Ganesan S, Bondioni MP, Ho ML, Marks C, Alajlan H, Mohammed RW, Zou F, Valencia CA, Filipovich AH, Facchetti F, Boisson B, Azzari C, Al-Saud BK, Al-Mousa H, Casanova JL, Abraham RS, Notarangelo LD. PAX1 is essential for development and function of the human thymus. Sci Immunol. 2020; 5(44): eaax1036.
- Xu W, Larbi A. Markers of T-cell senescence in humans. Int J Mol Sci. 2017; 18(8): 1742.
- Thewissen M, Somers V, Venken K, Linsen L, van Paassen P, Geusens P, Damoiseaux J, Stinissen P. Analyses of immunosenescent markers in patients with autoimmune disease. Clin Immunol. 2007; 123(2): 209-218.
- Staudacher O, Meyer T, Akbil B, Mayer M, Schmoll C, Kölsch U, Unterwalder N, Slagman A, Meisel C, Goffinet C, Möckel M, von Bernuth H. Autoantibodies against type I interferons correlate with low CD169/SIGLEC1 and severe non-viral infections in ER patients. Clin Exp Immunol. 2026; 220(1): uxaf074.
- Meisel C, Akbil B, Meyer T, Lankes E, Corman VM, Staudacher O, Unterwalder N, Kölsch U, Drosten C, Mall MA, Kallinich T, Schnabel D, Goffinet C, von Bernuth H. Mild COVID-19 despite autoantibodies against type I IFNs in autoimmune polyendocrine syndrome type 1. J Clin Invest. 2021; 131(14): e150867.
- Akbil B, Meyer T, Stubbemann P, Thibeault C, Staudacher O, Niemeyer D, Jansen J, Mühlemann B, Doehn J, Tabeling C, Nusshag C, Hirzel C, Sanchez DS, Nieters A, Lother A, Duerschmied D, Schallner N, Lieberum JN, August D, Rieg S, Falcone V, Hengel H, Kölsch U, Unterwalder N, Hübner RH, Jones TC, Suttorp N, Drosten C, Warnatz K, Spinetti T, Schefold JC, Dörner T, Sander LE, Corman VM, Merle U; Pa-COVID study Group; Kurth F, von Bernuth H, Meisel C, Goffinet C. Early and rapid identification of COVID-19 patients with neutralizing type I interferon auto-antibodies. J Clin Immunol. 2022; 42(6): 1111-1129.