Characterization of long-term immobility induced thromboprotective mechanisms in immune cells
Principle Investigator

Dr. Vincent Ehreiser
Scientific interest within the context of the graduate college
Disease prevention is taking a central role in tackling the burden of cardiovascular disease in our aging society. Thrombotic cardiovascular diseases such as venous thromboembolism, stroke and myocardial infarction are main drivers of morbidity and mortality worldwide. A central pathomechanism in these thrombotic cardiovascular diseases is termed thromboinflammation and arises from a deleterious dysregulation of an evolutionary conserved host defence mechanism involving platelets, the innate immune and coagulation system. There is an unmet clinical need for a better mechanistical understanding of thromboinflammation, in order to novel regulatory mechanisms that may pave the way for potential therapeutics, biomarkers and preventive measures. By pursuing this aim we established a across species approach to investigate the paradox that long-term immobility during hibernation in brown bears and patients with spinal cord injury (SCI) does not increase the risk of thrombosis. Our research acts at the interface between immunology, cardiovascular science and preventive medicine by taking advantage of an interdisciplinary translational research approach.
Project description
Cardiovascular diseases (CVDs), in particular myocardial infarction, stroke and venous thromboembolism (VTE), are the leading cause of morbidity and mortality worldwide, accounting for more than one third of all global deaths. Moreover, thrombotic events frequently and severely complicate a broad spectrum of clinical conditions that extend beyond the classical mechanisms of CVDs. Immobility and bed rest are among the strongest risk factors for VTE with increasing relevance in an aging population. Our previous study has demonstrated a naturally occurring thromboprotective phenotype in hibernating brown bears, bed-resting individuals, and SCI patients.1 This project examines neutrophils as central drivers of a thromboprotective phenotype during prolonged immobilization, employing a cross-species comparative approach that integrates hibernating brown bears and bed-resting human subjects. The overarching aim is to delineate thromboprotective signatures of neutrophils in long-term immobility, thereby uncovering neutrophil-intrinsic pathways that perpetuate thromboinflammatory responses. Building on these characterizations, we will establish the mechanistic foundation for identifying novel therapeutic targets in neutrophil-driven thromboinflammation.
Figure 1. Project scheme illustrative depiction of our cross-species approach to delineate thromboprotective signatures of neutrophils in immobility. Created with biorender.com.
In 2024 we serially captured free ranging brown bears during hibernation and the following activity period as previously described. Isolated neutrophils showed functional alterations, with reduced phagocytic activity reflected by decreased uptake of fluorescently labelled E. coli particles during winter. This indicates an altered neutrophil function between long‑term immobility (Figure 2/A-B), as well as reduced formation of neutrophil extracellular traps (NETs) (Figure 2/C). Extending our initial approach, we assessed neutrophil function in human volunteers undergoing a bed rest protocol at the DLR in Cologne. The functional profile reflects that observed in long‑term immobilized hibernating brown bears, with neutrophils displaying reduced phagocytotic capacities (Figure 1/D) and decreased NET formation in-vitro (Figure 2/E-F).
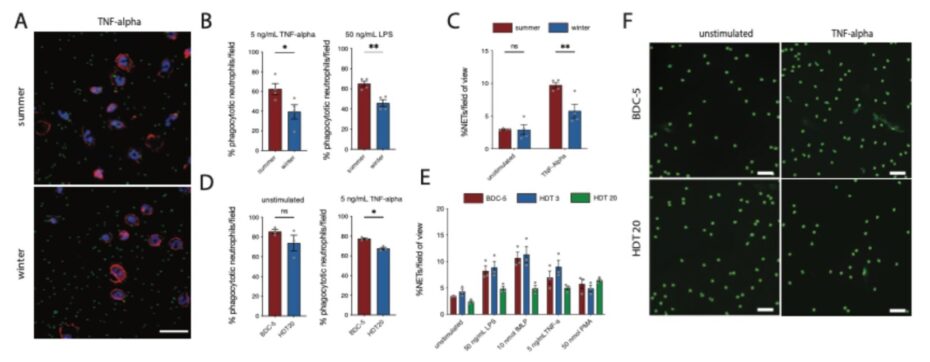
Figure 2. (A) Representative fluorescence microscopy images of neutrophils and labelled E. coli (scale bar: 20 µm). Phagocytosis and NET formation analysis. (B) Percentage of phagocytosing neutrophils in hibernating and active brown bears. (C) Comparative percentage of NET-forming neutrophils in hibernating brown bears. Comparative analysis in bed rest individuals at baseline mobility and after 20 days of bed rest, (D) percentages of phagocytosing and (E) NETing neutrophils. (F) Representative in vitro images of neutrophil NETosis (scale bar: 50 µm).
Quantitative proteomics (Figure 3) delineated differentially regulated proteins in brown bear neutrophils across summer activity and immobility during hibernation in winter. Concordantly, comparative proteomic analysis of neutrophils from our human bed-rest cohort demonstrated immobility-associated downregulation of the ARP2/3 complex as well as associated downstream proteins. The ARP2/3 complex is a key component of the actin cytoskeleton, and its inhibition has previously been shown to impair cellular migration in neutrophils.2-4 Moreover, neutrophils derived from patients with rare inherited defects in actin polymerization, specifically due to deficiencies in the actin-related protein 2/3 complex exhibited a defect for NETosis.5
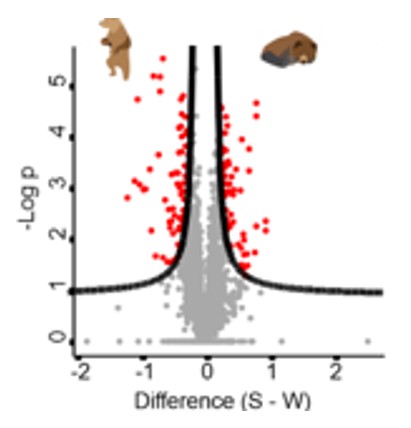
Figure 3. Quantitative proteomic analysis of hibernating and active brown bears. Blue marking for significant proteins with a p-value <0.01.
Aim 1: Identification of neutrophil phenotypes associated with thromboprotection in two models of immobility across species. We hypothesize that neutrophil functions, encompassing phagocytosis, NETosis, actin-dependent spreading, migration, and degranulation are reprogrammed during prolonged immobility in hibernation in brown bears, resulting in a functionally hyporesponsive phenotype in neutrophils that attenuates prothrombotic potential and confers protection against immobility-associated VTE. Within Aim 1, we will delineate these functional alterations through an established collaboration with the Scandinavian Brown Bear Research Project procuring neutrophils from hibernating and active bears in Sweden for on-site in-vitro experiments and validate our previous results. Building on our proteomic analyses, which predominantly identified proteins of the actin‑associated cytoskeleton, we will specifically assess neutrophil migration and cell‑spreading behavior.6 In addition, during the active phase of the brown bears, neutrophils will be treated in vitro with an Arp2/3 complex inhibitor to assess whether their prothrombotic phenotype under conditions of mobility can be shifted toward an antithrombotic phenotype. In analogy to the experiments described in brown bears, we will conduct in vitro experiments as well as further studies within the collaboration of the bed rest study. In analogy to the experiments described in brown bears, we will conduct in vitro experiments as well as further studies within the collaboration of the bed rest study. Cell-free supernatants derived from neutrophil stimulation experiments will be used for proteomic analysis to quantify secreted mediators, including myeloperoxidase (MPO) and neutrophil elastase, thereby correlating degranulation dynamics with antithrombotic phenotype during hibernation. Plasma samples will be subjected to immunoprofiling by ELISA and mass spectrometry-based cytokine arrays to identify pro-inflammatory and thrombogenic mediators, such as interleukins and other DAMPs implicated in neutrophil priming and NETosis in mobility.
Aim 2: Neutrophil crosstalk reveals thromboprotective phenotypes enabling therapeutic targets in immobility-related thromboinflammation. In Aim 2, we will dissect differentially regulated proteins and signalling pathways identified in neutrophils from hibernating brown bears and bed-resting individuals, assessing their capacity to evade short-term immobility-induced proinflammatory phenotypes and promote a thromboprotective state under chronic immobilization. Building on our preliminary data we identified the ARP2/3 complex as a prominently downregulated protein during immobility in neutrophils from hibernating brown bears and human bed-resting individuals. Thus, we will perform functional in-vitro experiments to assess its effect on neutrophil phenotypes, and its influence on the interaction between neutrophils and immune cells (i.e. platelets, monocytes) in the context of thromboinflammation. To this end, we will treat isolated neutrophils with ARP 2/3 inhibitors or vehicle controls and subsequently subject them to phenotypic and functional analysis to assess neutrophil effector functions. Neutrophil functionality will be evaluated using established phagocytosis assays, NETosis quantification, as well as degranulation assays. Additionally, co-culture models (i.e. platelet-neutrophils) will be employed to quantify ARP 2/3 complex inhibition effects on NET release under thromboinflammatory conditions, and we will furthermore assess neutrophil migration in-vitro. In addition, experiments in specific transgenic MRP8-CreArp2/3-deficient mice are planned, which will provide further insights by employing the inferior vena cava flow‑restriction model that induces venous thrombosis and thereby allowing the visualization of thrombus formation and its modulation by targeted interventions.7
References
- Thienel M, Müller-Reif JB, Zhang Z, Ehreiser V, Huth J, Shchurovska K, Kilani B, Schweizer L, Geyer PE, Zwiebel M, Novotny J, Lüsebrink E, Little G, Orban M, Nicolai L, El Nemr S, Titova A, Spannagl M, Kindberg J, Evans AL, Mach O, Vogel M, Tiedt S, Ormanns S, Kessler B, Dueck A, Friebe A, Jørgensen PG, Majzoub-Altweck M, Blutke A, Polzin A, Stark K, Kääb S, Maier D, Gibbins JM, Limper U, Frobert O, Mann M, Massberg S, Petzold T. Immobility-associated thromboprotection is conserved across mammalian species from bear to human. Science. 2023; 380(6641): 178-187.
- Henson JH, Yeterian M, Weeks RM, Medrano AE, Brown BL, Geist HL, Pais MD, Oldenbourg R, Shuster CB. Arp2/3 complex inhibition radically alters lamellipodial actin architecture, suspended cell shape, and the cell spreading process. Mol Biol Cell. 2015; 26(5): 887-900.
- Glaser KM, Doon-Ralls J, Walters N, Rima XY, Rambold AS, Réategui E, Lämmermann T. Arp2/3 complex and the pentose phosphate pathway regulate late phases of neutrophil swarming. iScience. 2024; 27(1): 108656.
- Fritz-Laylin LK, Riel-Mehan M, Chen BC, Lord SJ, Goddard TD, Ferrin TE, Nicholson-Dykstra SM, Higgs H, Johnson GT, Betzig E, Mullins RD. Actin-based protrusions of migrating neutrophils are intrinsically lamellar and facilitate direction changes. Elife. 2017; 6: e26990.
- Sprenkeler EGG, Tool ATJ, Henriet SSV, van Bruggen R, Kuijpers TW. Formation of neutrophil extracellular traps requires actin cytoskeleton rearrangements. Blood. 2022; 139(21): 3166-3180.
- Gaertner F, Ahmad Z, Rosenberger G, Fan S, Nicolai L, Busch B, Yavuz G, Luckner M, Ishikawa-Ankerhold H, Hennel R, Benechet A, Lorenz M, Chandraratne S, Schubert I, Helmer S, Striednig B, Stark K, Janko M, Böttcher RT, Verschoor A, Leon C, Gachet C, Gudermann T, Mederos Y Schnitzler M, Pincus Z, Iannacone M, Haas R, Wanner G, Lauber K, Sixt M, Massberg S. Migrating platelets are mechano-scavengers that collect and bundle bacteria. Cell. 2017; 171(6): 1368-1382.e1323.
- von Brühl ML, Stark K, Steinhart A, Chandraratne S, Konrad I, Lorenz M, Khandoga A, Tirniceriu A, Coletti R, Köllnberger M, Byrne RA, Laitinen I, Walch A, Brill A, Pfeiler S, Manukyan D, Braun S, Lange P, Riegger J, Ware J, Eckart A, Haidari S, Rudelius M, Schulz C, Echtler K, Brinkmann V, Schwaiger M, Preissner KT, Wagner DD, Mackman N, Engelmann B, Massberg S. Monocytes, neutrophils, and platelets cooperate to initiate and propagate venous thrombosis in mice in vivo. J Exl Med. 2012; 209(4): 819-835.